INTRODUCTION
In the production of gene therapies, viral vectors are commonly utilized in bioprocessing to introduce modified genetic material into host cells. Immortalized Human Embryonic Kidney (HEK) cells are frequently used due to transfection efficiency as well as cell growth in applications such as vaccine manufacturing and process intensification. Typical viral vector processes consist of the associated vector with the modified DNA present binding to the cell membrane where the vector becomes packaged in the cell’s vesicle. From there, said vesicle continues to break down the releasing factor into the cell, and the cell starts making new proteins utilizing the new gene introduced.
When collecting the valuable phase (either the cells or supernatant) that contains the transfected virus, different separation methods have been utilized, such as depth filtration, tangential flow filtration, and centrifugation. When attempting to separate the virus like particles from the supernatant with depth filtration and tangential flow filtration, lower recovery rates and suboptimal turbidity readings have been found to impact cell collection for downstream processing. This case study presents data from the CARR Biosystems team highlighting the UniFuge family and the ability to clarify HEK293 cells in a manner yielding better results than other forms of filtration.
THE CHALLENGE
Manufacturers of gene therapies continue to implement new solutions to optimize their existing process of collecting virus like particles through clarifying HEK293 cells. Frequent challenges for viral vector production include high costs, poor filtration, and inefficient recovery. Common methods of analyzing for successful clarification are testing for associated Nephelometric Turbidity Units (NTU), cell viability, and cell recovery. When attempting to collect the targeted cells for further processing, it is essential for therapeutic developers to implement a process that yields a clarified supernatant while maintaining high recovery with minimal impact to viability throughout the entire process.
THE SOLUTION
The introduction of the scalable UniFuge family provides manufacturers the ability to obtain efficient results with the clarification of HEK293 cells in various stages of production from research to manufacturing. Through low shear separations at different G-forces, UniFuge users are able to acquire optimal recovery and viability rates at all stages of the production process, ranging from research and development to manufacturing. When comparing to traditional filtration processes, bioprocessors can trust that the UniFuge family will continue performing reliably through scale-up activity while maintaining a closed system from start to finish.
THE RESULTS
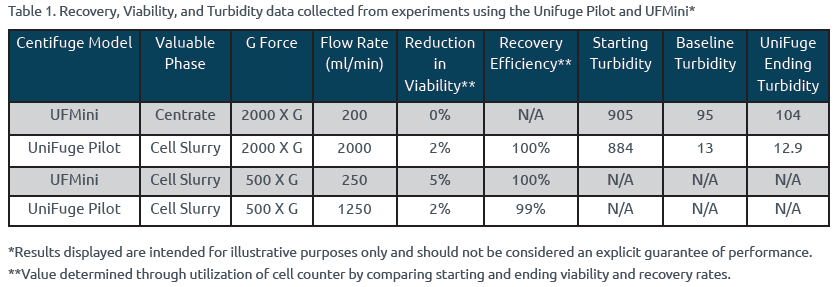
The UniFuge Pilot and UFMini have demonstrated high yield and viability rates while also maintaining target NTU readings. Extensive studies have proven that the UniFuge family has minimal impact on recovery rates and turbidity readings, indicating high performance over a wide range of operating conditions used in gene therapy processes. Table 1 presents examples of viral vector applications which showcase significant reductions in turbidity as well as strong recovery and viability rates.
DISCUSSION
As presented in Table 1, whether the customer is looking to clarify centrate or the cell slurry, the UniFuge Pilot and UFMini have demonstrated minimal impact to viability, maximized recovery, and decreased turbidity readings in comparison to other filtration methods.
The performance results of the UniFuge Pilot and UFMini with viral vector applications provide bioprocessing experts with confidence that cell separation can be achieved in an aseptic and effective manner for further research and manufacturing processes.
ABOUT THE UNIFUGE FAMILY
The UniFuge family provides low-shear separation, high-recovery performance, and fast processing times in three scalable models: UFMini, UniFuge Pilot, and U2k. With a variety of bowl sizes and flow rates ranging from 29 ml/min to 20 L/min, the UniFuge family of single-use separation systems offer both scalability and process efficiency in an aseptic closed system. The UniFuge Pilot was the industry pioneer in single-use centrifuge separation technologies. In 2021 CARR Biosystems released the UFMini to better support customers with a need for smaller scale processing using single-use technology
ABOUT CARR BIOSYSTEMS
Founded in 1993 in Medfield, Massachusetts, CARR Centritech introduced the Powerfuge and continued to develop advanced separation systems for the Life Sciences and Specialty Chemical markets with the Viafuge, Centritech, and UniFuge models. In 2022, CARR Centritech transitioned to CARR Biosystems with continued emphasis on developing high-quality products to meet safety and quality standards throughout a variety of applications across the globe.